Medwatch 25. september 2024
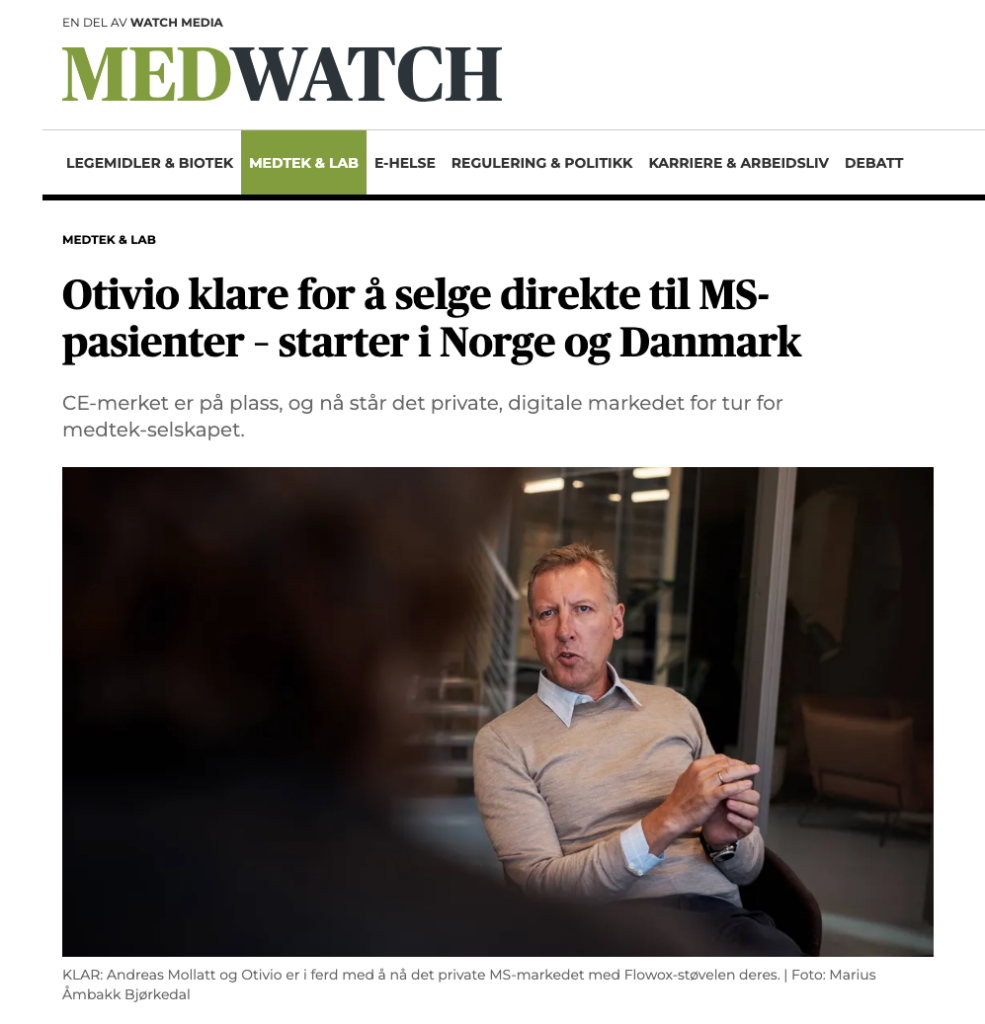
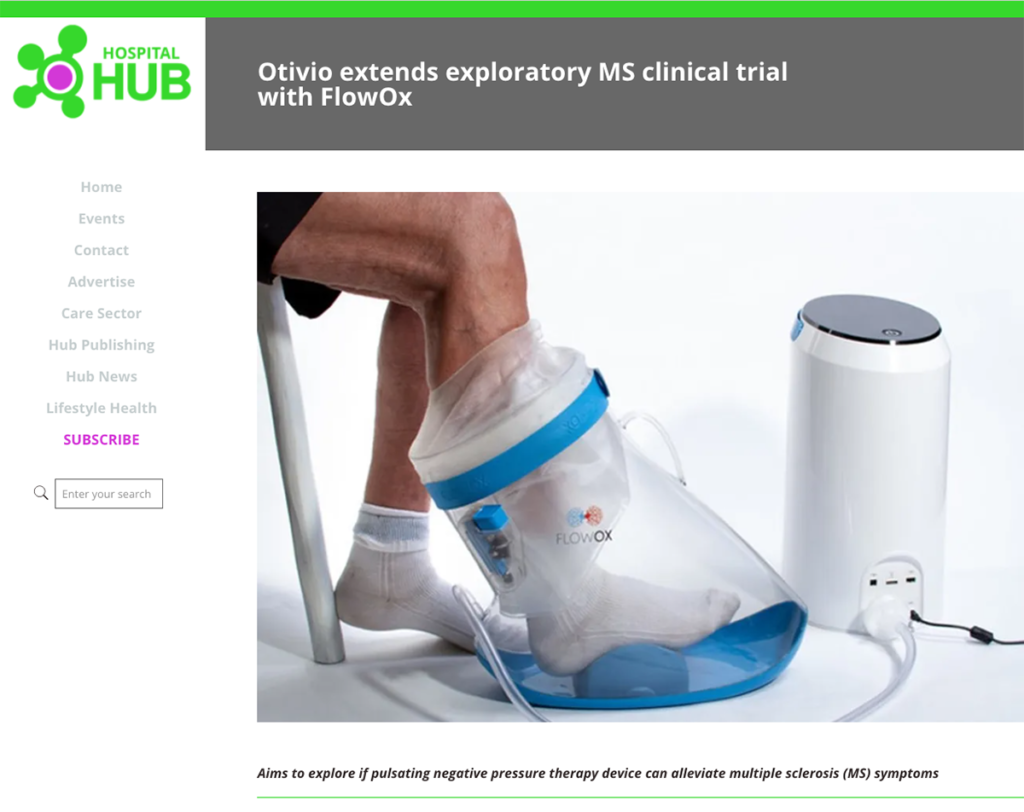
Medtech company Otivio is on a strong trajectory as it moves toward commercializing its pressure boot, Flowox, designed for multiple sclerosis (MS) patients. The company recently received CE marking for Flowox, indicating its approval for use with MS patients. Furthermore, promising results have been noted regarding the treatment of spasticity in MS patients.
Medwatch 25. september 2024 Read More »