FlowOx™ by Otivio
Otivio is a small medtech development company based in Oslo, Norway. Our vision is to stimulate active self care and treatment through simple and easy to use home treatment devices and services.
BIOGRAPHY
FlowOx™ by Otivio is a new direction in self healthcare
Otivio AS is a privately owned company based in Oslo, Norway. The company was founded in 2010 based on IP, prototypes and clinical data developed at University of Oslo. Otivio is ISO 13485 certified. Our main product FlowOx™ is a non-invasive medical device for home use addressing insufficient blood flow to the limbs associated with peripheral arterial disease, diabetes and other medical conditions. These conditions lead to pain, chronic wounds and amputations.
FlowOx™ is documented to improve these outcomes which have a big impact on Quality of Life and health care costs. FlowOx™ represents a solution for transition from hospital to home treatment accentuated by the COVID-19 outbreak. FlowOx™ is CE marked (CE2460) and available in numerous countries.

EMPLOYEES
Who are we at Otivio?
The management team of Otivio has, on average, more than 20 years of experience in the life science industry.
ABOUT FLOWOX™
FlowOx™ in the Press
Otivio has developed FlowOx™ for patients suffering from the effects of PAD such as leg pain and chronic wounds. Early long-term testing documents increased blood flow.
Otivio raised NOK 40 million.
Article in Annals of Vascular Surgery
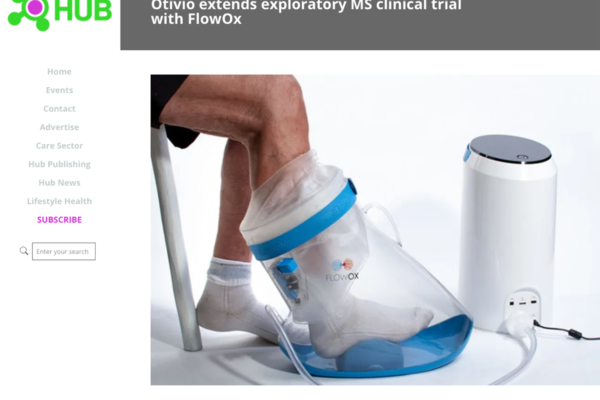
Article in Hospital Hub
Article in Health Tech World
Medtech article on Otivio’s MS Discovery
First RCT to show improved walking capacity
COOPERATIVE PARTNERS
With a little help
Otivio has a strong setup of partners and suppliers all the way from design, development and manufacturing to clinical documentation, sales and distribution.

Special Plast
Gislaved, Sweden
Specialplast has been producing MedTech devices in and outside clean room since 1995. We are a quality focused subcontractor who develops and produces items in close cooperation with our customers.
NEWS
What’s new with FlowOx™?
Here are our latest news regarding Otivio and FlowOx™.